How to calculate the boiling point of a mixture made up of 2 liquids ( acetone+water in the same ratios) - Quora
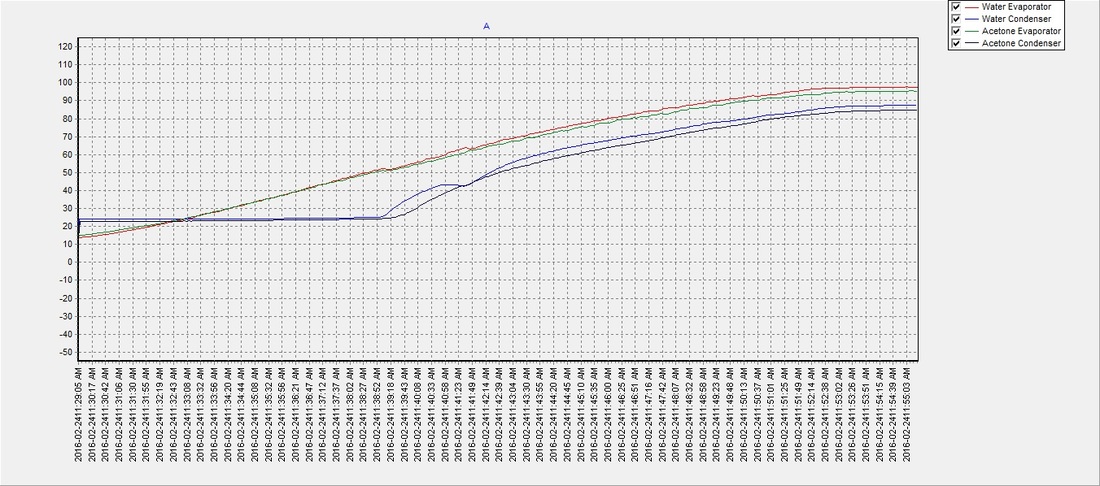
Simple Distillation 120 100 80 60 40 20 0 0 5 10 15 20 25 30 35 40 2. Using the graph calculate the boiling points of ac - Answer Happy

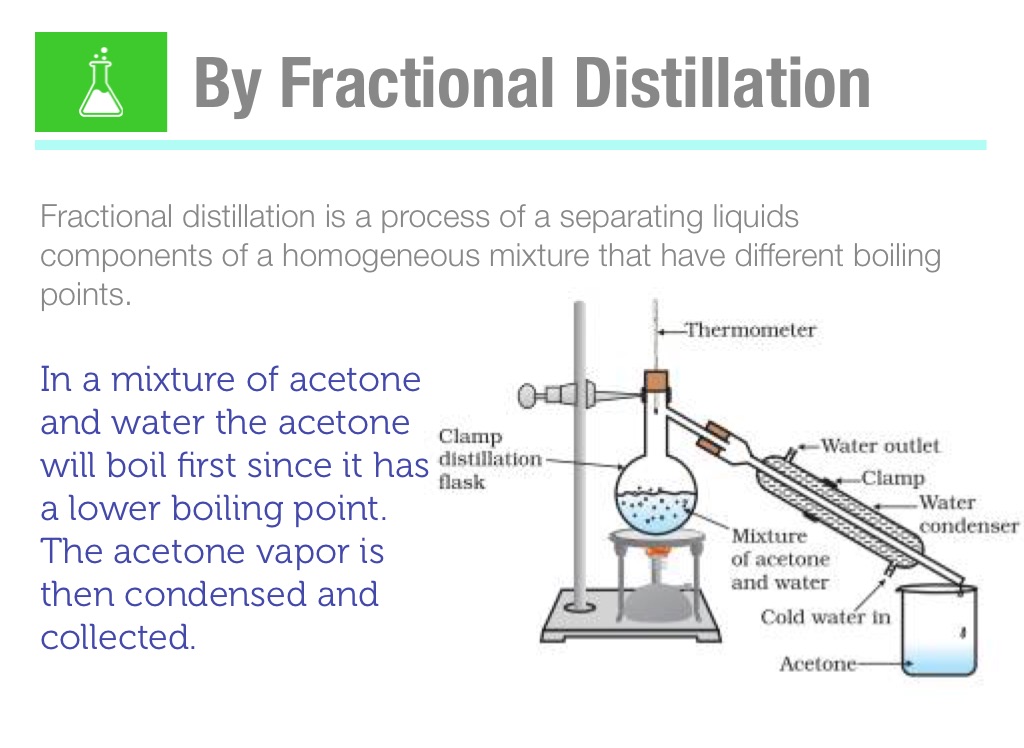
DOC) Chem 120 Laboratory VOLATILITY: DISTILLATION AND DETERMINATION OF BOILING POINT | Ian capuno - Academia.edu

The boiling point of pure acetone is 56.38^(@)C. When 0.707 g of a compound is dissolved in 10 g of acetone there is elevation to 56.88^(@)C in b.p What is the mol.wt.

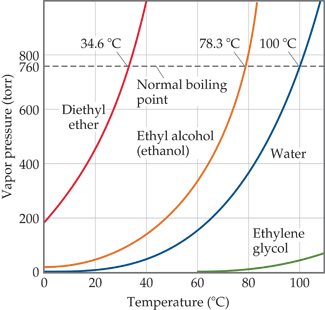
filosoffen.dk - what is metformin 500 mg used for | Congratulate, this acetone boiling point graph you inquisitive