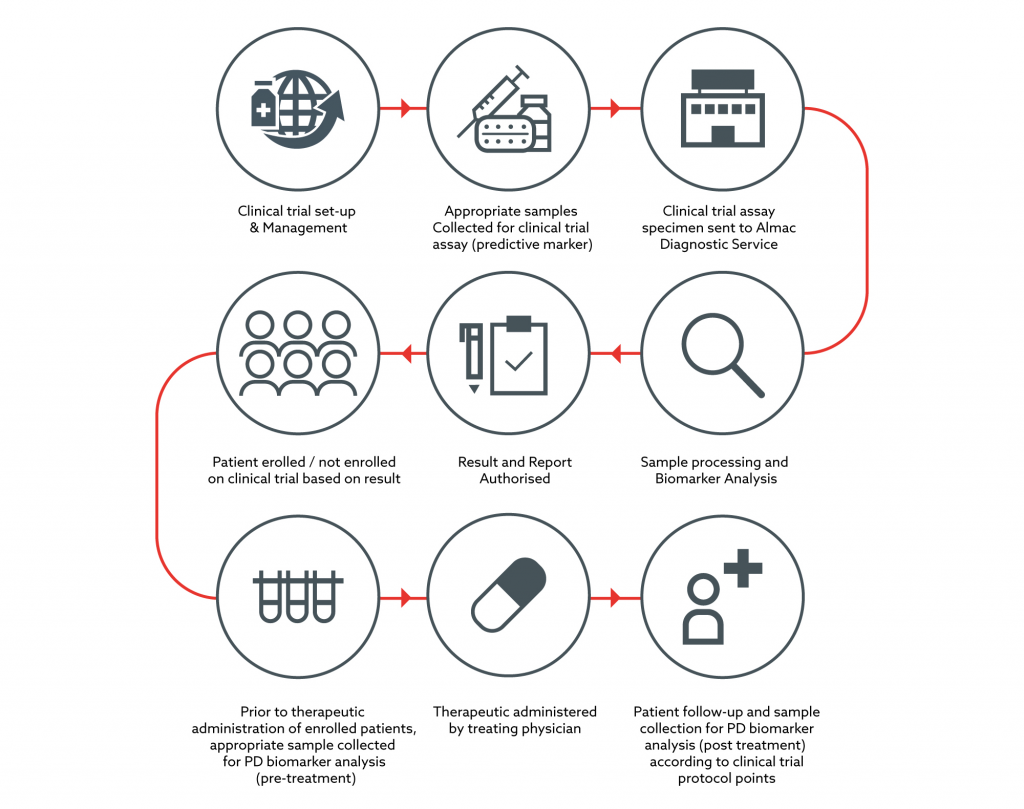
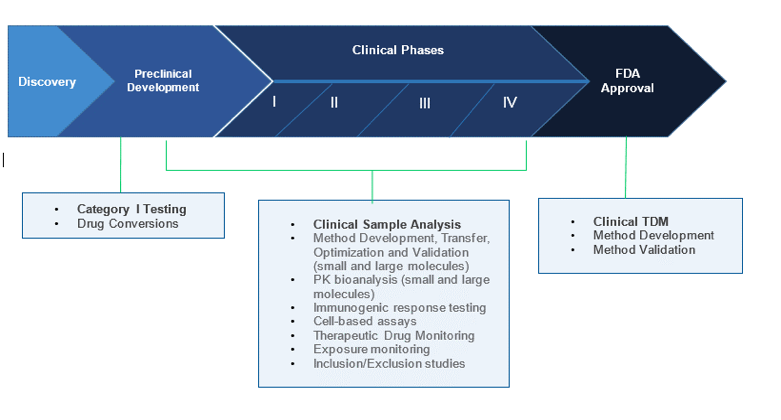
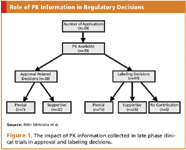
Pharmacokinetic and Statistical Considerations in First-in-Human Clinical Trials | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services
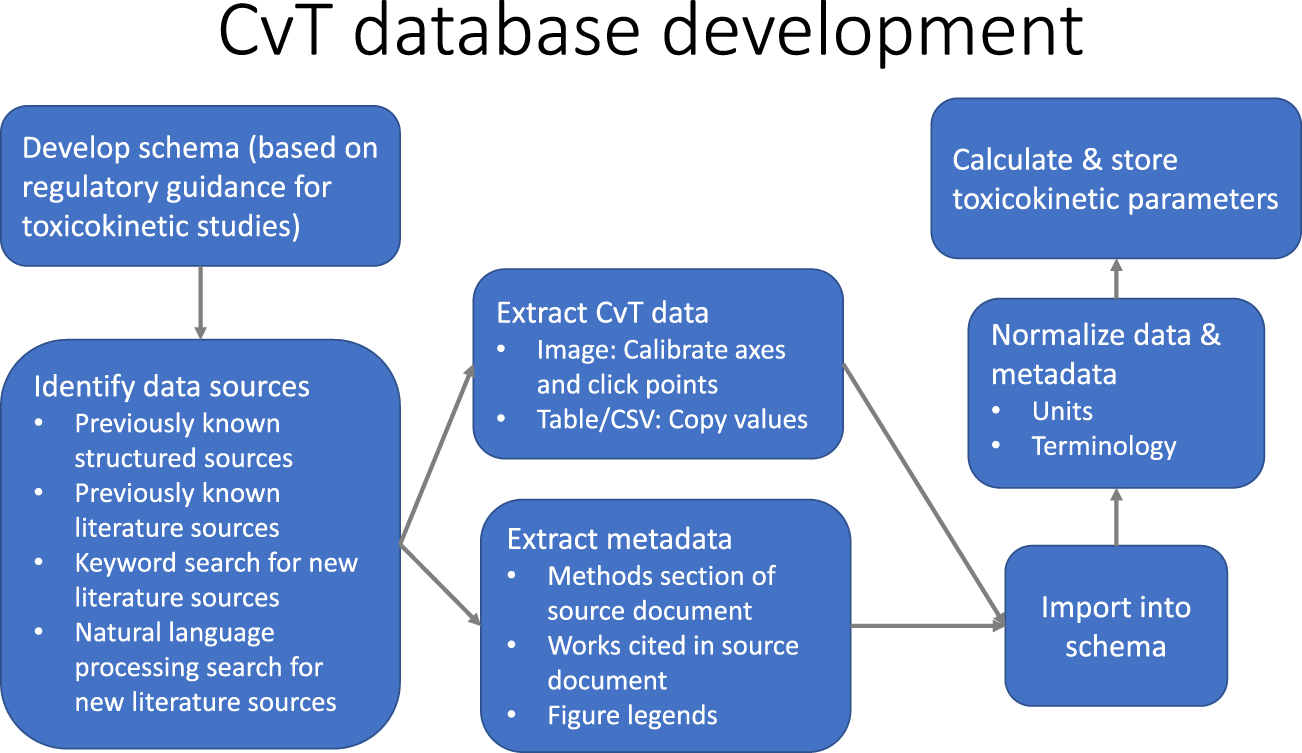
Database of pharmacokinetic time-series data and parameters for 144 environmental chemicals | Scientific Data

Phase 1 clinical trial schema. Abbreviations: E = enrollment, qwk = per... | Download Scientific Diagram

Dose rationale and pharmacokinetics of dexmedetomidine in mechanically ventilated new-borns: impact of design optimisation | SpringerLink
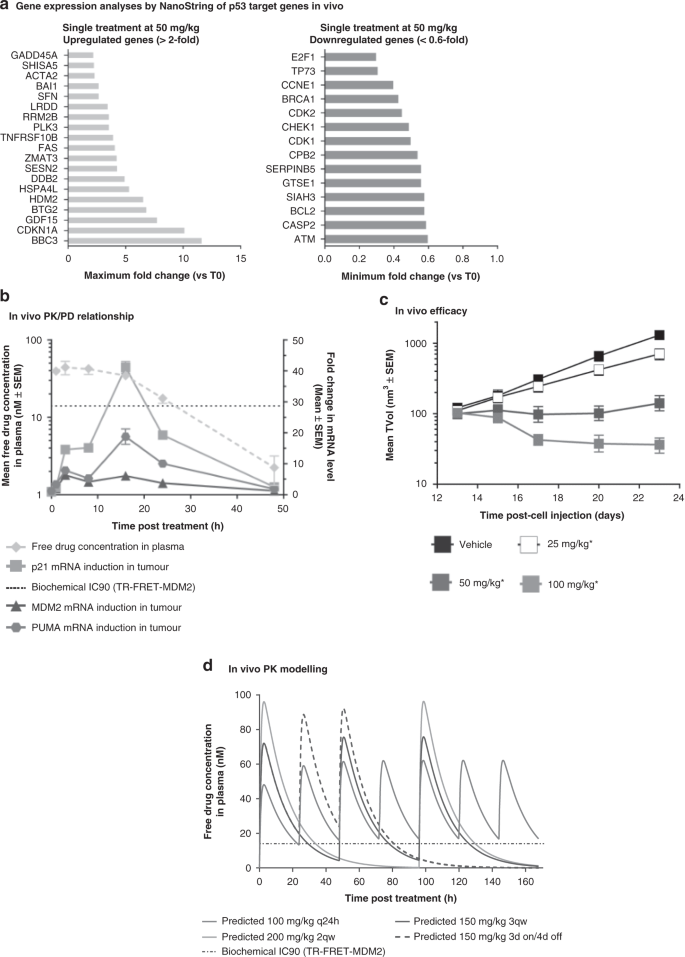
Pharmacokinetic–pharmacodynamic guided optimisation of dose and schedule of CGM097, an HDM2 inhibitor, in preclinical and clinical studies | British Journal of Cancer
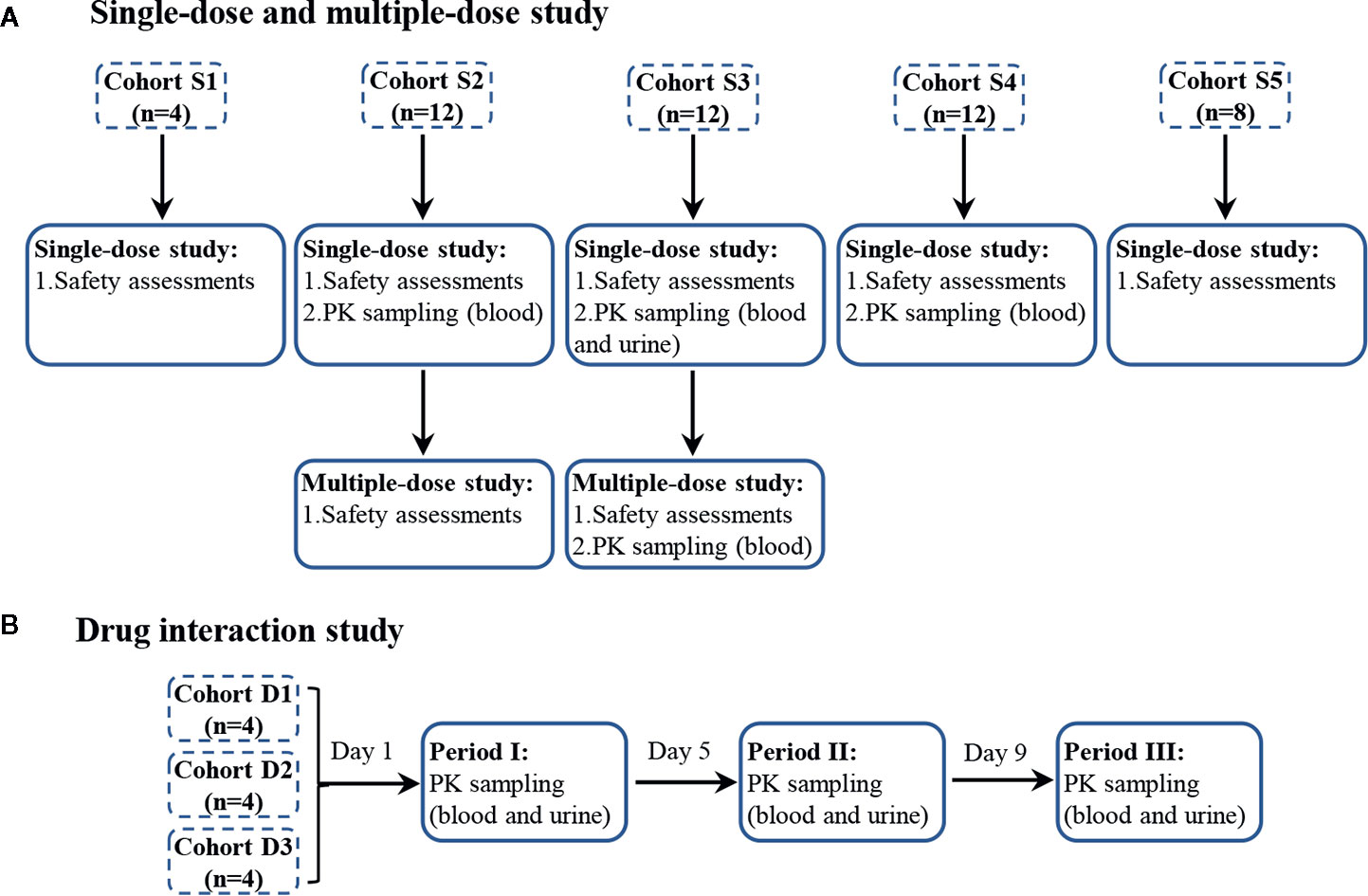
Frontiers | Tolerability, Safety, Pharmacokinetics and Drug Interaction of Cefotaxime Sodium–Tazobactam Sodium Injection (6:1) Following Single and Multiple Intravenous Doses in Chinese Healthy Subjects | Pharmacology

Pharmacokinetic and Statistical Considerations in First-in-Human Clinical Trials | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

Phase 0 clinical trials in oncology new drug development Gupta UC, Bhatia S, Garg A, Sharma A, Choudhary V - Perspect Clin Res

Pharmaceutics | Free Full-Text | Pharmacokinetic-Pharmacodynamic Modelling of Systemic IL13 Blockade by Monoclonal Antibody Therapy: A Free Assay Disguised as Total | HTML

Pharmacokinetic and Statistical Considerations in First-in-Human Clinical Trials | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

Power estimation using a population pharmacokinetics model with optimal design by clinical trial simulations: application in pharmacokinetic drug– drug interaction studies | Vidho Rivera - Academia.edu

How to design a study to evaluate therapeutic drug monitoring in infectious diseases? - Clinical Microbiology and Infection

a Staggered approach and sampling strategy for collecting PK/PD and... | Download Scientific Diagram

On Biostatistics and Clinical Trials: Serial Blood Sample Timepoints for Comparing Pharmacokinetics Profiles Between Two Different Dose Frequencies

![PDF] Phase 0 Clinical Trial Strategies for the Neurosurgical Oncologist | Semantic Scholar PDF] Phase 0 Clinical Trial Strategies for the Neurosurgical Oncologist | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ba643ff4c798a7864e6a78f009413a89e764a2c0/6-Figure2-1.png)