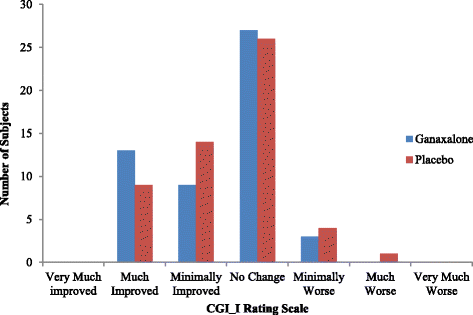
A randomized double-blind, placebo-controlled trial of ganaxolone in children and adolescents with fragile X syndrome | Journal of Neurodevelopmental Disorders | Full Text

Safety and efficacy of ganaxolone in patients with CDKL5 deficiency disorder: results from the double-blind phase of a randomised, placebo- controlled, phase 3 trial - The Lancet Neurology

Safety and efficacy of ganaxolone in patients with CDKL5 deficiency disorder: results from the double-blind phase of a randomised, placebo- controlled, phase 3 trial - The Lancet Neurology

Ganaxolone Fragile X Clinical Trial Showed Disappointing Results • Fragile X Research - FRAXA Research Foundation
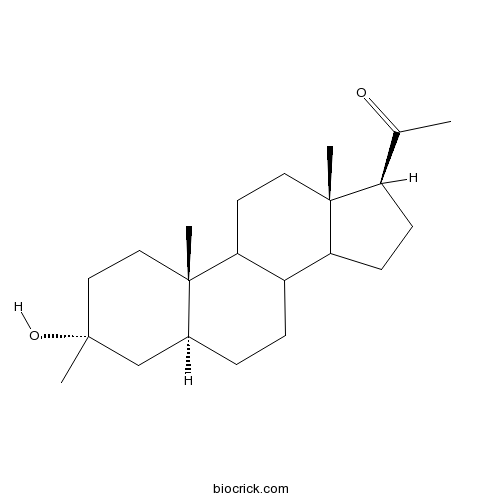
Ganaxolone | CAS:38398-32-2 | Potent, positive allosteric modulator of GABAA receptors | High Purity | Manufacturer BioCrick

Trial design. GNX-GNX group: randomized to ganaxolone for the first... | Download Scientific Diagram

Randomized, double‐blind, placebo‐controlled phase 2 study of ganaxolone as add‐on therapy in adults with uncontrolled partial‐onset seizures - Sperling - 2017 - Epilepsia - Wiley Online Library

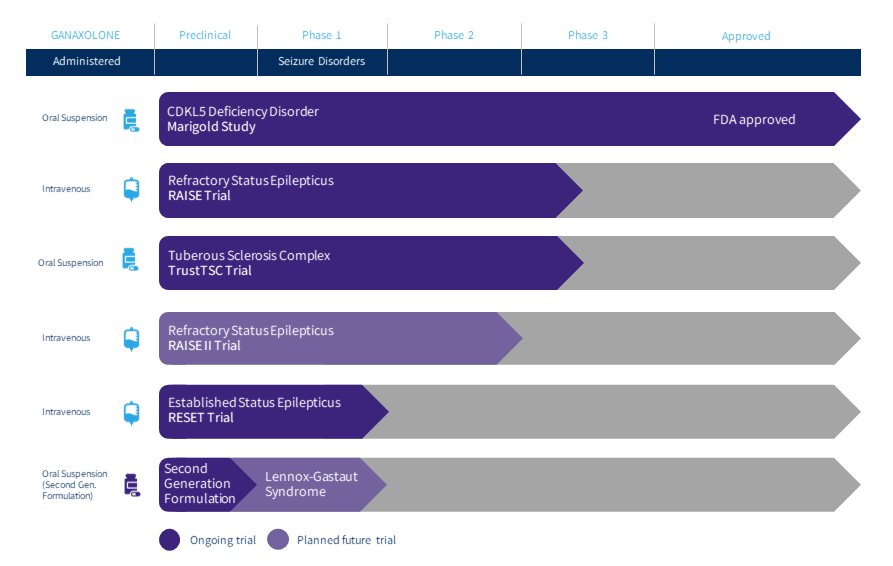
Marinus Pharmaceuticals Announces Delay to RAISE Phase 3 Clinical Trial in Status Epilepticus and Associated IV Ganaxolone Clinical Trials | Business Wire









![Ganaxolone stereoselective inhibition of [ 35 S]TBPS binding | Download Table Ganaxolone stereoselective inhibition of [ 35 S]TBPS binding | Download Table](https://www.researchgate.net/profile/Richard-Carter-4/publication/14145484/figure/tbl1/AS:601618521280538@1520448441976/Ganaxolone-stereoselective-inhibition-of-35-STBPS-binding.png)
