![PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/62bcc7885dac29517026f3ba577045d3b93dbb7b/2-Table2-1.png)
PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar

Discontinuation and non-publication of surgical randomised controlled trials: observational study | The BMJ

Do clinical trials reflect reality? A systematic review of inclusion/exclusion criteria in trials of renal transplant immunosuppression - Ayaz‐Shah - 2018 - Transplant International - Wiley Online Library

Eligibility Criteria and Representativeness of Randomized Clinical Trials That Include Infants Born Extremely Premature: A Systematic Review - The Journal of Pediatrics

Enhancing pediatric clinical trial feasibility through the use of Bayesian statistics. – Pubrica Academy

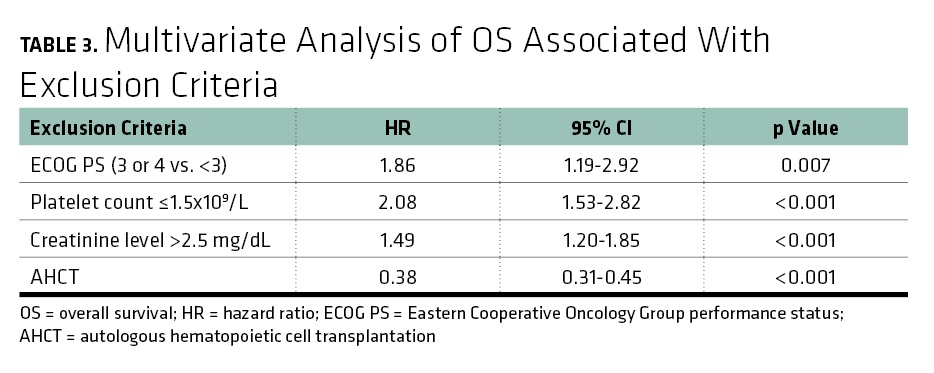
Re-Evaluating Eligibility Criteria for Oncology Clinical Trials: Analysis of Investigational New Drug Applications in 2015 | Journal of Clinical Oncology